Findings may lead to improved treatment and prevention of atherosclerosis and the formation of blood clots
A study led by researchers at St. Joseph’s Healthcare Hamilton and McMaster University has found a significant link between the immune system and its role in the acceleration of plaque build-up in blood vessels. This discovery represents a promising advancement in the field of cardiovascular health that may lead to improved treatments for atherosclerosis (plaque build-up inside the blood vessels) and thrombosis (blood clots inside a blood vessel).
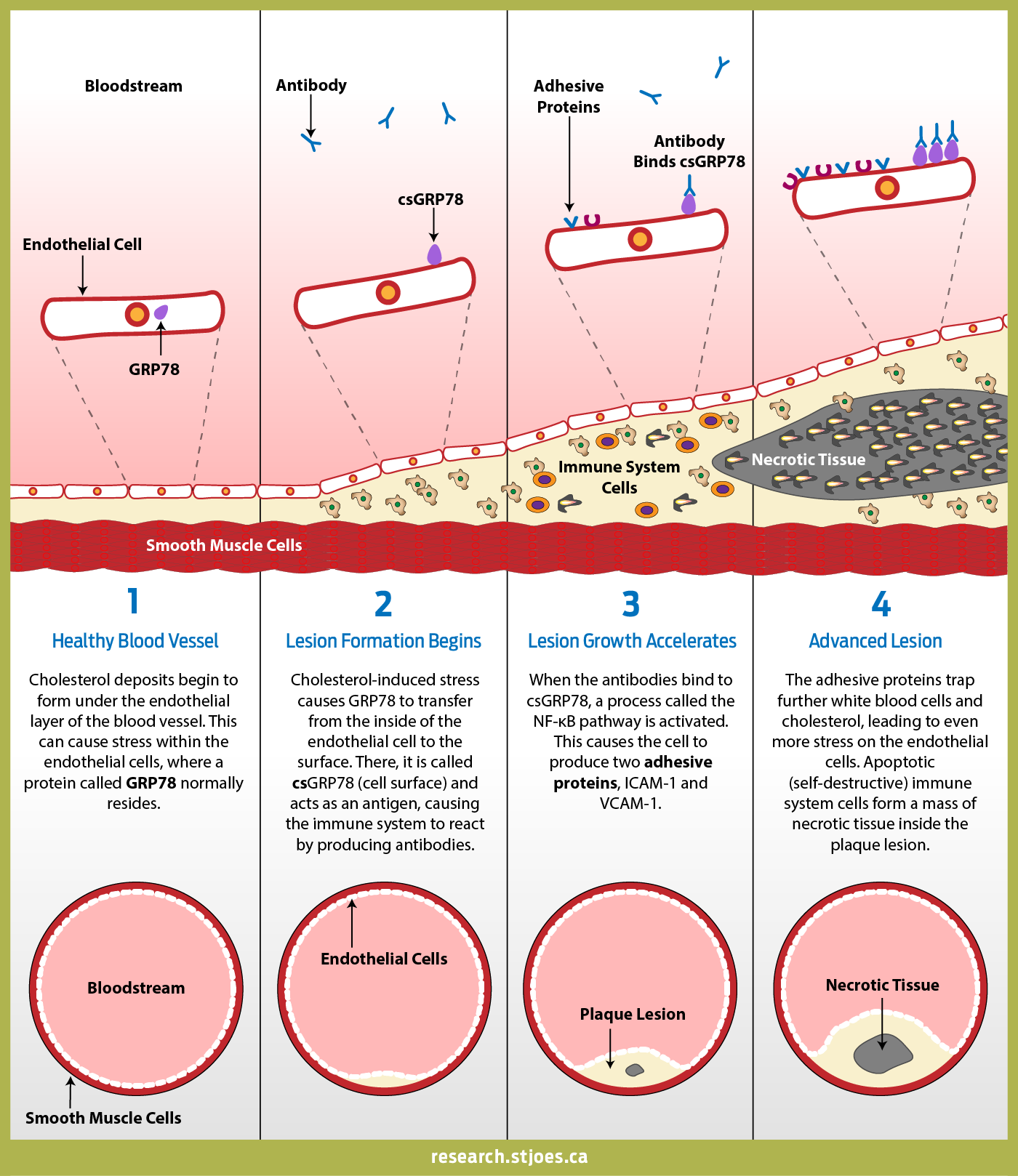
Atherogenesis is the process by which the build-up of plaque – sometimes called lesions – form just below the endothelial cells that line the inside of blood vessels. Plaque build-up is a step towards various cardiovascular problems, including stroke, heart attack, aneurism, and more. Atherosclerosis is the leading cause of cardiovascular disease in the developed world.
Previous research has shown that various forms of circulating cholesterol, such as LDL (commonly considered the “bad” cholesterol), can cause the endothelial cells to become stressed. This stress is a known hallmark of atherosclerotic lesion development.
Researchers have now uncovered a specific cellular mechanism that causes accelerated lesion growth. They found that when endothelial cells experience stress caused by the build-up of cholesterol in plaque, a protein called GRP78 is transported to the surface of these cells.
Inside the cell, GRP78 normally functions to assist in protein synthesis and folding. Under stress conditions, including high levels of bad cholesterol, GRP78 can appear on the cell surface to initiate a domino effect that activates the immune system and accelerates plaque development. This has led researchers to theorize that atherosclerosis could be an autoimmune disease.
“The immune system is essentially recognizing the presence of GRP78 on the cell surface and attacking it by generating antibodies. When this occurs, a complex process leads to the appearance of two adhesive proteins,” explained Dr. Ali Al-Hashimi, co-first author of the study. “These adhesive proteins act to recruit certain immune cells (termed monocytes) that contribute to the formation of larger, less stable plaques. This can ultimately lead to plaque rupture and the formation of life-threatening blood clots.” Al-Hashimi is a postdoctoral fellow in the laboratory of Dr. Austin at St. Joseph’s Healthcare Hamilton.
The researchers also found that enoxaparin, a form of the blood-thinning drug heparin, was able to block the interaction between cell surface GRP78 and the antibodies in an atherosclerosis-prone mouse model. This effectively halts the cellular domino effect that otherwise leads to accelerated plaque development.

Dr. Richard Austin (left) and Dr. Ali Al-Hashimi (right) in the Austin Research Lab at St. Joe's
Additional studies by Drs. Al-Hashimi, Shayegan and Austin at St. Joseph’s Healthcare Hamilton have shown that enoxaparin was effective at reducing GRP78’s role in accelerating prostate tumour growth.
“The observation that the FDA-approved and commercially available drug enoxaparin (Lovenox) may protect against atherosclerotic lesion growth and may stabilize the plaque…could well open up new therapeutic strategies,” said Dr. Arap, director of Rutgers Cancer Institute of New Jersey in Newark.
“Now that we have a clearer understanding of the complex process that links these antibodies to cell surface GRP78 and accelerates lesion build-up, we can identify new compounds/drugs that disrupt this cell surface GRP78/antibody interaction,” said Dr. Richard Austin, senior author of the study. Dr. Austin is a Professor in the Division of Nephrology at McMaster University, a Career Investigator of the Heart and Stoke Foundation of Canada and the Amgen Canada Research Chair in Nephrology at St. Joseph’s Healthcare Hamilton.
In addition, the study demonstrated that patients diagnosed with cardiovascular disease have significantly elevated blood levels of GRP78 antibodies. This suggests that this process can occur in cardiovascular patients, supporting the findings of a link between the immune system and lesion growth.
The study was a collaborative effort between the Austin Lab at The Research Institute of St. Joe’s – Hamilton, Dr. Wadih Arap and Dr. Renata Pasqualini from the Rutgers Cancer Institute of New Jersey, and Dr. Katey Rayner from the University of Ottawa. It is the first study to demonstrate the actual mechanisms behind GRP78, cellular stress, and its role in atherogenesis.
The study was published in the Journal of Clinical Investigation Insight, a peer-reviewed, open access publication. Funding was provided by the Canadian Institutes of Health Research and the Heart and Stroke Foundation of Canada. Support from the Research Institute of St. Joe’s was also provided.
As a leading nephrology research centre, St. Joseph’s Healthcare Hamilton conducts investigations on the cellular mechanisms involved in atherosclerosis, as well as other areas of vascular and kidney health.